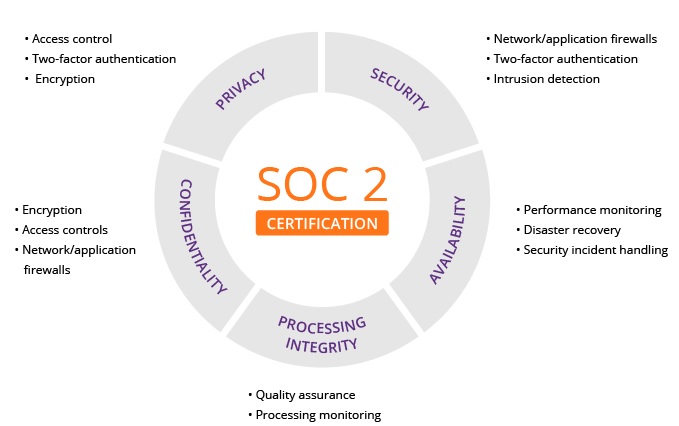
FDA CFR Part 11 Compliance
VirtualTitle 21 CFR Part 11 is the part of Title 21 code of Federal Regulations that establish the Food and Drug Administration (FDA) regulations on electronic records and electronic signatures (ERES). Part 11 applies to records in electronic form that are created, modified, maintained, archived, retrieved, or transmitted under any records requirements set forth in Agency regulations. Part 11 also applies […]