Security Risk Analysis for Meaningful Use, MIPS/MACRA and HIPAA
Our certified privacy and security professionals can help your organization comply with the requirements in an efficient and cost-effective way. Or, simply leverage our DIY toolkit to complete the risk assessment and meet the MIPS/MACRA requirements in less than an hour.
Why Does Security Risk Analysis for Meaningful Use, MIPS/MACRA and HIPAA Matter?
Protecting patient health information is a base score measure for MIPS/MACRA eligible clinicians to receive positive or downward payment adjustments in 2019. Any organization that is a Covered Entity or Business Associate under HIPAA regulations MUST also complete an annual security risk assessment, then maintain a supporting risk management plan for a potential HHS/OCR audit. In addition, NCQA PCMH program provides 2 credits (TC5) recognition for completing an annual security risk assessment.

Why databrackets?
- Online Do-it-Yourself Security Risk Analysis Toolkit
- Experienced and Certified Consultants
- Audit Support Guarantee
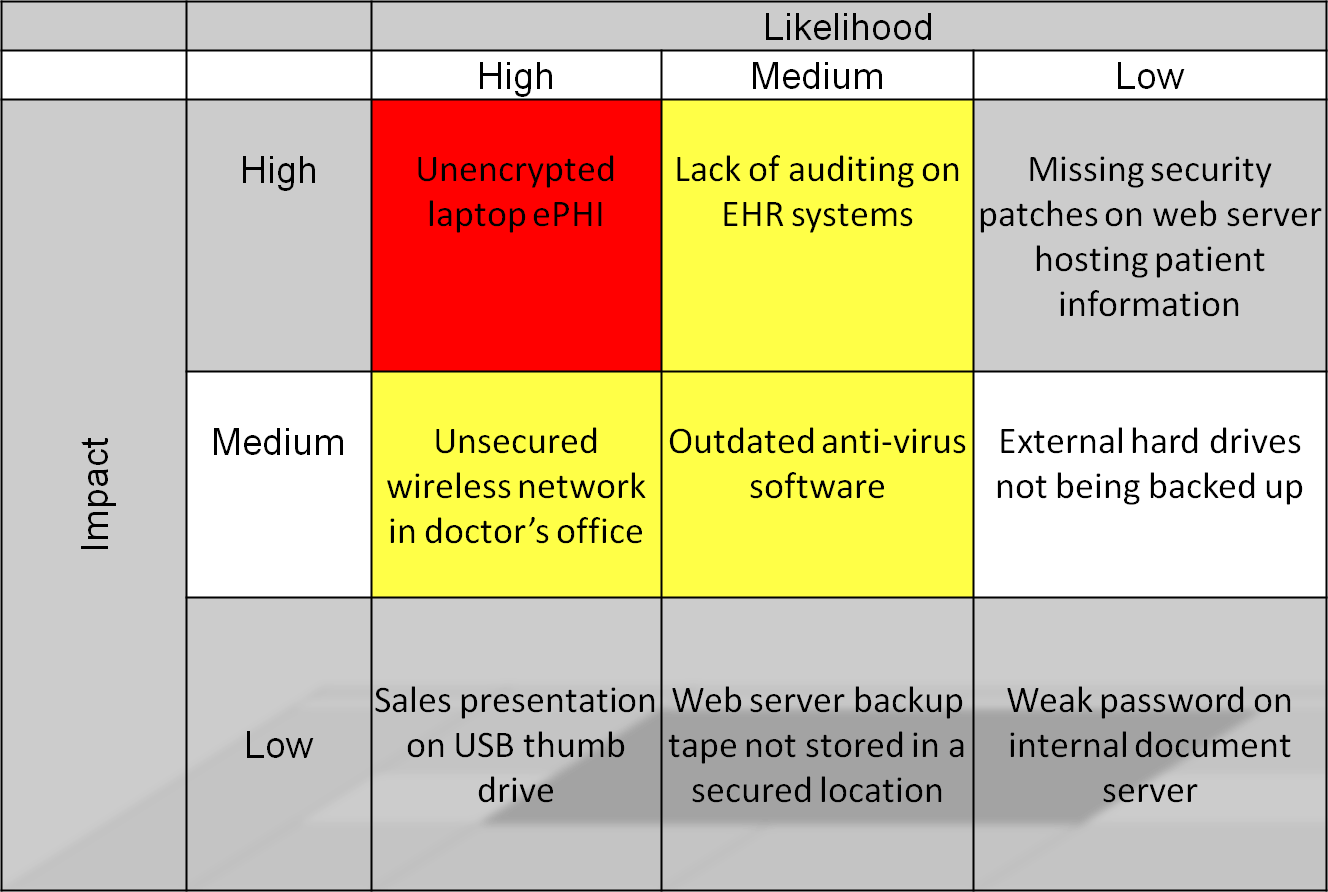
Prioritized Security Risks
Our Deliverables
- Defining the scope of the security risk analysis
- Inventorying ePHI (electronic Protected Health Information) systems
- Reviewing past security risk assessment report(s)
- Assessing current and trending security measures
- Determining the likelihood of threat occurrence
- Identifying risks using automated and manual analysis
- Prioritizing implementation/remediation plans
- Documentation of findings
- Security risk assessment report
- Executive summary
- Results summary
- Remediation plans
- Detail report based on individual assessment areas
- HIPAA security awareness training
- Customized policies and procedures
- Optional continuous compliance plans
Interested in trying our DIY platform ?
Security Risk Analysis Showcase
Additional Resources
Also, the visitors of our website have an option to use the SRA tool provided by ONC/NIST. Please read their disclaimer section thoroughly. In addition, the free tool doesn’t include network assessment, training and policies, and procedures. Ensure you complete a comprehensive security risk analysis and audit-proof your report. Our toolkit has been designed to help busy medical professionals like you to complete the risk assessment in less than an hour.













